Key Takeaways
- Hemp Industry Regulations:
- Decree No. 372/014 regulates hemp for non-medical purposes.
- Resolution 82/020 regulates hemp for medical purposes.
- Authorizations for cultivation, processing, sale, import and export of hemp intended for non-medical use are issued by the Ministry of Livestock, Agriculture and Fisheries.
- Licenses that allow the cultivation, processing and sale of hemp for medical use are issued by the Regulation and Control of Cannabis.
- Obtaining an authorization for import or export of hemp for medical purposes follows a Single Window for Foreign Trade (VUCE) procedure.
- Uruguay Hemp CBD Industry Stats:
- In 2024, 37.6 tons of hemp for medical purposes were cultivated in Uruguay: a 55.5% increase from 2023.
- Nearly 23,000 kilograms of hemp were exported from Uruguay in 2023 which accounts for nearly 95% of total hemp production in that year.
Hemp Legalization
Uruguay legalized industrial hemp in 2013 through Law 19.172. Under the country’s law, hemp is defined to include plants or part of plants of the cannabis genus that have no more than 1% THC and seeds of different varieties of hemp that have no more than 0.5% THC. Industrial hemp was then regulated in 2014 by Decree No. 372/014 which made the Ministry of Livestock, Agriculture and Fisheries (MGAP) responsible for authorizing non-medicinal hemp-related activities and the licensing process of hemp businesses. The General Directorate of Agricultural Services (DGSA), which is under the MGAP, is responsible for evaluating and authorizing businesses to operate with hemp.
Through Resolution 82/020, in 2020 the authority for licensing the cultivation and post-harvest activities relating to hemp for medicinal purposes was given to the Regulation and Control of Cannabis (IRCCA).
Hemp Licenses Available in Uruguay
To perform activities such as cultivation, processing, marketing, import and export of hemp intended for non-medical use, an authorization from the MGAP must be obtained. To perform activities with hemp intended for medical use, such as for consumption by people or animals or for use in cosmetics, a license from the IRCCA must be obtained.
Non-Medicinal Purposes
Fees:
Cultivation for flower harvest and high CBD feminized seeds.
0-5 hectares or 0-600 meters squared: free of charge
5-20 hectares or 601-1200 meters squared: 1,000 Uruguayan Indexed Units
20-50 hectares or 1,201-2,500 meters squared: 2,500 Uruguayan Indexed Units
more than 50 hectares or more than 2,500 meters squared: 4,500 Uruguayan Indexed Units
Cultivation for grains, seeds for agricultural uses or stem biomass.
0-100 hectares: free of charge
101-500 hectares: 1,000 Uruguayan Indexed Units
more than 500 hectares: 2,500 Uruguayan Indexed Units
The authorization for cultivation and post-harvest activities relating to hemp is valid for one year. Import authorizations expire 120 days after their issuance while export authorizations expire after 90 days. The import and export authorizations can be used only once.
Licensing process:
- Applicants must first register in the Single Operators Registry of the DGSA, designate their technical manager(s). Applicants wishing to provide seed germination or seedling cutting services must also be registered nurseries in the Single Operators Registry.
- The applicant must appoint their Agricultural Engineer. The individual chosen to be an Agricultural Engineer must be registered with the Single Operators Registry as a technician.
- A report from SENACLAFT will also be required.
- Then applicants must submit a request to the Director of the DGSA, signed by the legal representative of the applicant and the Agricultural Engineer responsible for the production process, with a work plan which must contain the following information:
- Identification of the applicant.
- The activities the applicant intends to perform with hemp.
- Site where planting, cultivation and harvesting of hemp will take place.
- Origin of the seeds and plants that will be used.
- Varietal characteristics of the crops that will be used.
- THC content in plants and seeds.
- Safety procedures that will be applied.
- Destination of production and crop residues.
- Characteristics of the final product.
- Applicants who receive the authorization will then be registered in the Cannabis Registry under the IRCCA.
In case of an export authorization:
Before an export authorization is granted, the applicant must present an import certificate issued by the responsible authorities of the importing country, authorizing the import.
In case of hemp production for use in veterinary medicines:
The General Directorate of Livestock Services must grant the authorizations for the industrial plants that extract cannabis resins and cannabinoid isolates for veterinary medical use. The drying of the raw hemp material that is intended to be used in the veterinary pharmaceutical industry must be carried out in establishments authorized by the Department of Medicines or the Food Safety and Quality Directorate of the DGSA.
More details on the application process and what information must be submitted to obtain an authorization can be found in the following guide from the MGAP.
Medicinal Purposes
Fees:
Category 1: 45,000 Indexed Units
Category 2: 60,000 Indexed Units
Category 3: 120,000 Indexed Units
Category 4: 240,000 Indexed Units
Seed production: 45,000 Indexed Units
Category 1: 45,000 Indexed Units
Category 2: 60,000 Indexed Units
Category 3: 120,000 Indexed Units
Category 4: 240,000 Indexed Units
Criteria for each category:
| Category 1 | Category 2 | Category 3 | Category 4 | ||
| Maximum number of flowering plants at any one time | Indoor/Greenhouse | Up to 2,500 | 2,501-5,000 | 5,001-10,000 | Over 10,000 |
| Maximum number of vegetative plants at any one time | Indoor/Greenhouse | Up to 2,500 | 2,501-5,000 | 5,001-10,000 | Over 10,000 |
| Kilograms of plant material per year | Both (indoor/greenhouse and outdoor) | Up to 1,500 | 1,501-5,000 | 5,001-25,000 | Over 25,000 |
| Cultivation area (hectares) | Outdoor | Up to 2.0 | 2.1-5.0 | 5.1-25.0 | Over 25.0 |
Licensing process:
- Submit a request for a license with all the required information to the IRCCA.
- The IRCCA’s technical team will evaluate the request and once the evaluation process is completed and a favorable report from SENACLAFT is received, the request will be submitted to the IRCCA Board of Directors.
- The Board of Directors will decide on whether to grant the license and will establish the conditions of the license.
Information required to be submitted is not limited to:
- The objective of the hemp-related activity and purposes for which production will be used.
- If applying for a cultivation authorization:
- The origin and quantity of the starting material.
- Varietal characteristics of the material to be used with their certificates of analysis showing the cannabinoid content of each variety.
- Description of the production process.
- Traceability control procedure.
- Surplus, waste and derived by-product management procedure.
- Stock control procedure.
- Layout of facilities.
- Analytical specifications for the release of finished products.
- Description of the geographical location of the proposed hemp establishment.
- Activities that will be outsourced.
- Cash flow forecast.
- Income Statement forecast, on an annual basis.
- Operating and security measures.
Obtaining an authorization for import or export of hemp for medical purposes follows a Single Window for Foreign Trade (VUCE) procedure. There is a published guide on how to use VUCE for importing or exporting hemp in Uruguay.
Renewal of Authorizations and Licenses
The authorization for cultivation and post-harvest activities relating to hemp received from the MGAP must be renewed every year. The registered hemp establishment with an authorization from the MGAP must submit its work plan for the next year and a notarial certificate stating that there have been no changes in its corporate structure.
Hemp establishments with a license from the IRCCA must renew their license every year and pay the relevant license fee annually.
Uruguayan Hemp CBD Industry
In September 2021, according to Uruguay XXI (Uruguay’s Investment, Export and Country Brand Promotion Agency), there were 76 companies growing hemp in Uruguay on 456 hectares in the open field. A November 2024 report from Uruguay XXI reveals that in 2023 there were 35 companies authorized by the MGAP to grow hemp on 335 hectares in outdoor production and on 43,000 square meters in indoor facilities. Among the licensees, 22 were growing hemp flower on 41 hectares outdoors and on 31,690 square meters indoors while the 4 licensees growing for hemp grain, cultivated hemp on 173 hectares outdoors. This demonstrates that companies producing hemp for hemp flower prefer to do so in controlled environments. Around 55% of the hemp grown outdoors in Uruguay is for industrial purposes such as fiber and grain.
In 2023, a total of 28.6 tons of medical cannabis was produced and more than 80% of it was non-psychoactive cannabis – hemp for medical purposes. In 2024, the amount of hemp cultivated for medical purposes rose to 37.6 tons: a 55.5% increase from 2023.
Nearly 23,000 kilograms of hemp were exported from Uruguay in 2023 which accounts for nearly 95% of total hemp production in that year. This demonstrates that the hemp produced in Uruguay is mainly destined for export. Hemp seeds were exported mainly to Argentina (11,561kg) with the rest being exported to Costa Rica, Spain and Paraguay. In 2022, Switzerland was the main destination of Uruguay’s hemp flowers with US$834,887 worth of hemp flowers exported to the country. The amount of hemp flower exports fell from 13,284kg in 2022 to 11,356kg in 2023. Switzerland remained as the major receiver of the exported hemp flower (6,407kg), followed by the Czech Republic (2,701kg) and the U.S. (1,628kg).
Uruguay Hemp CBD Industry Infographics
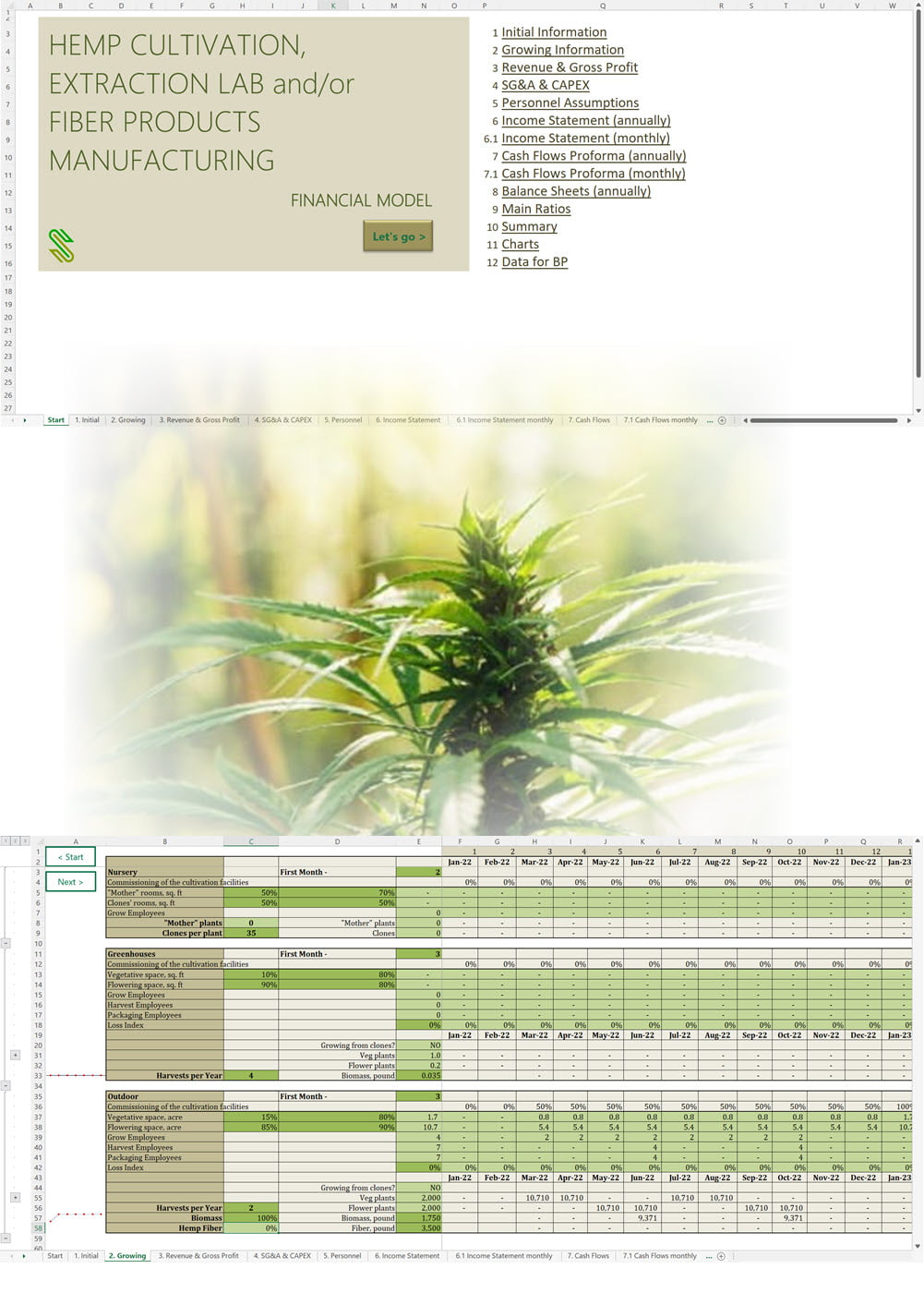
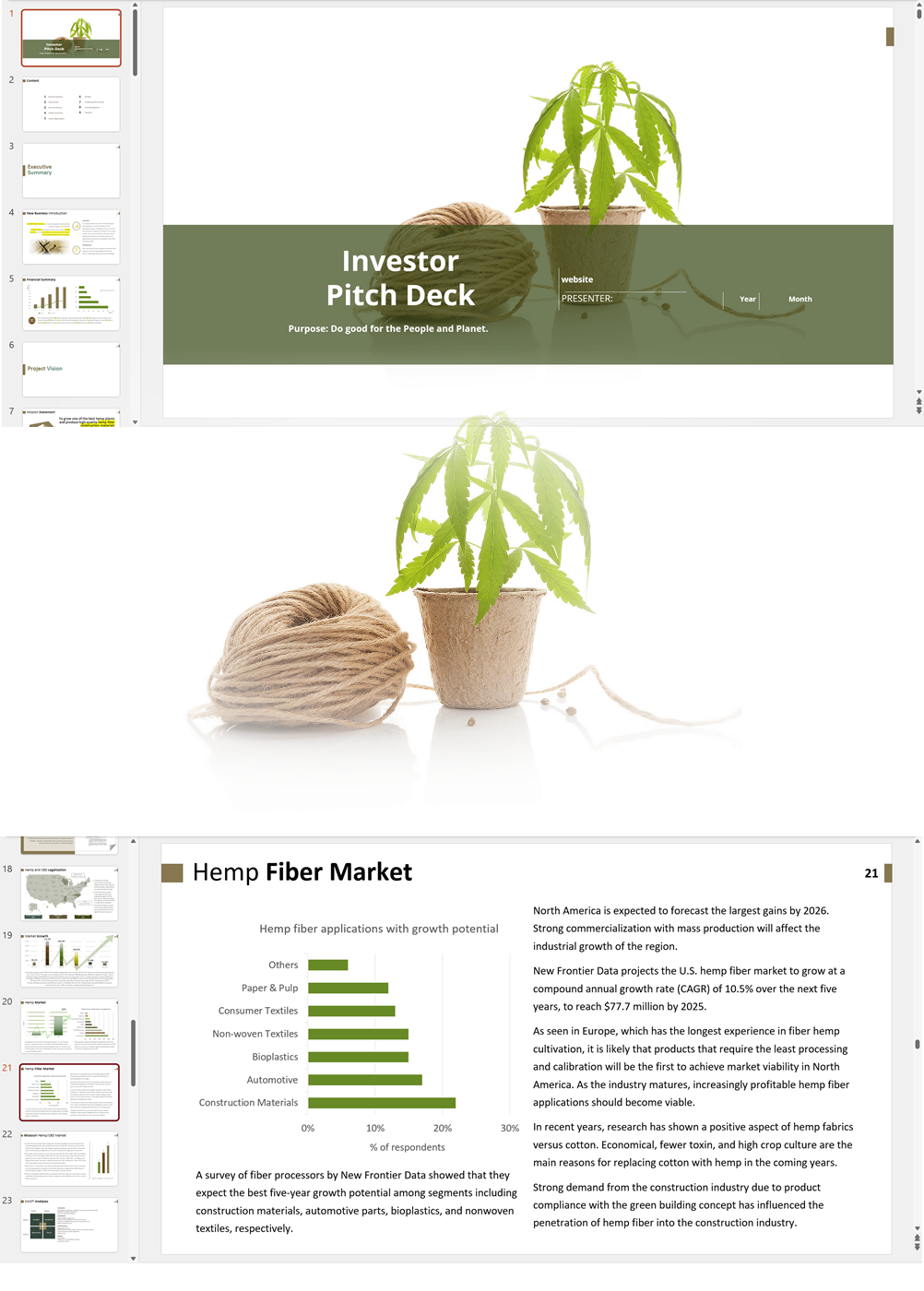
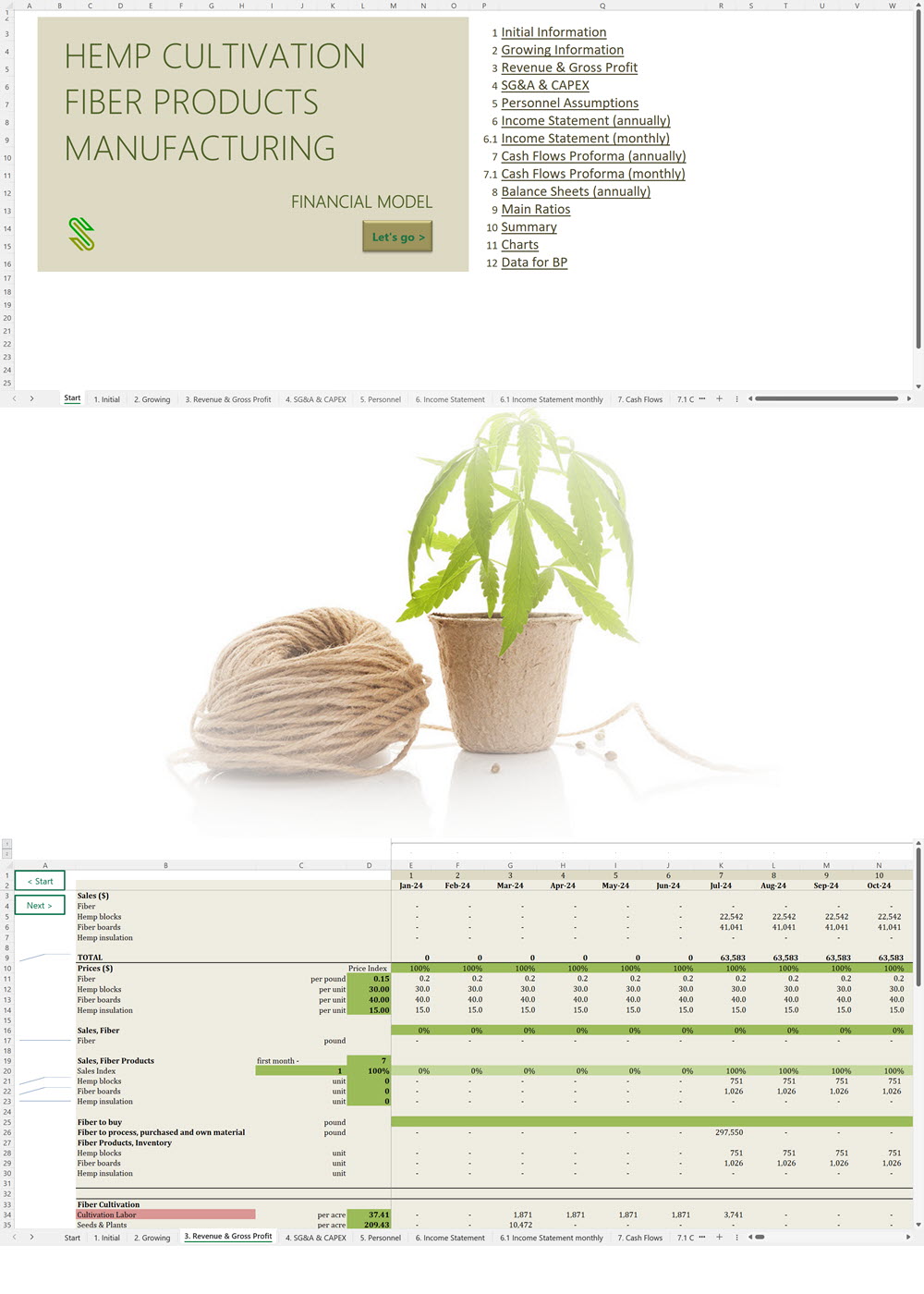
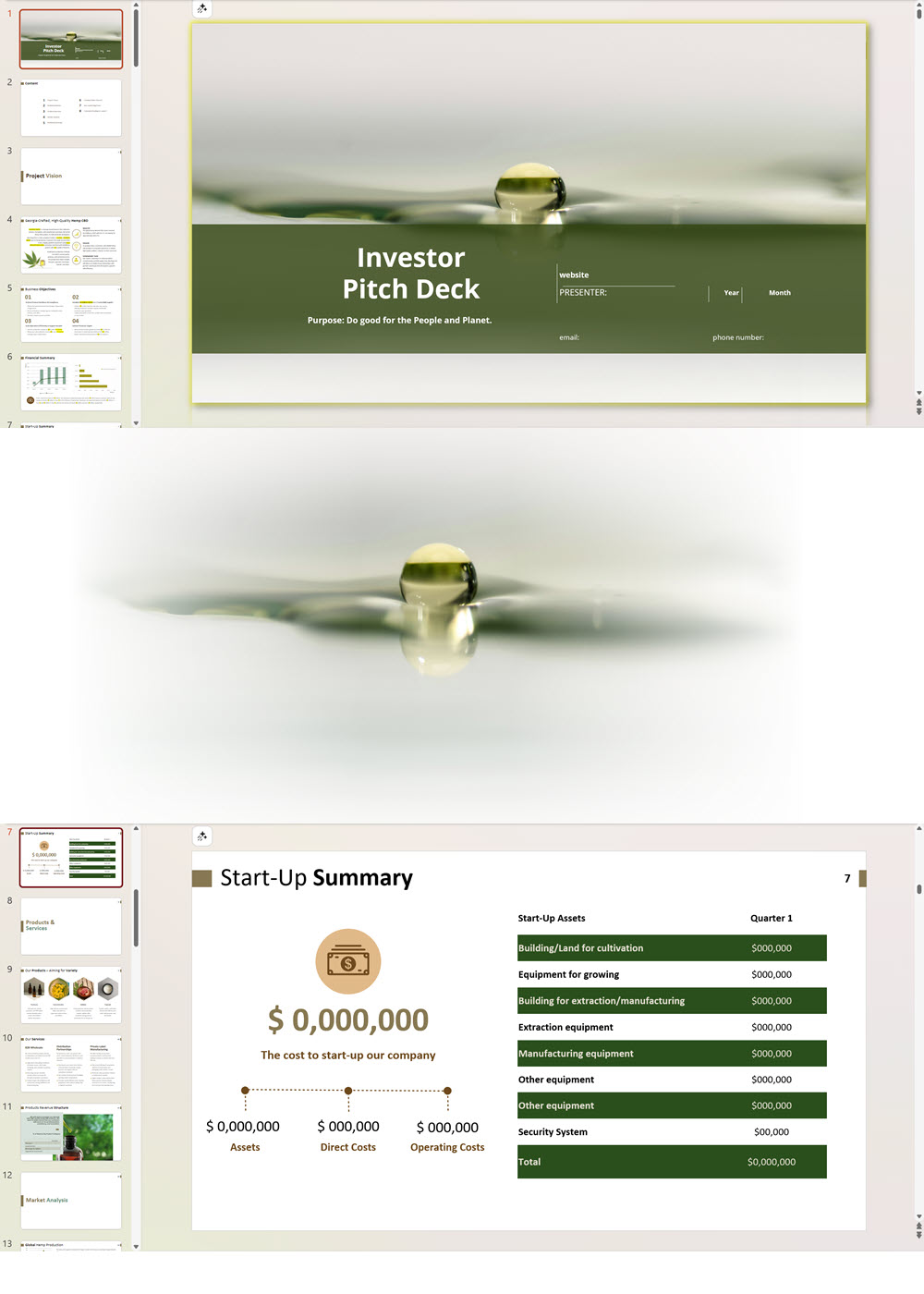
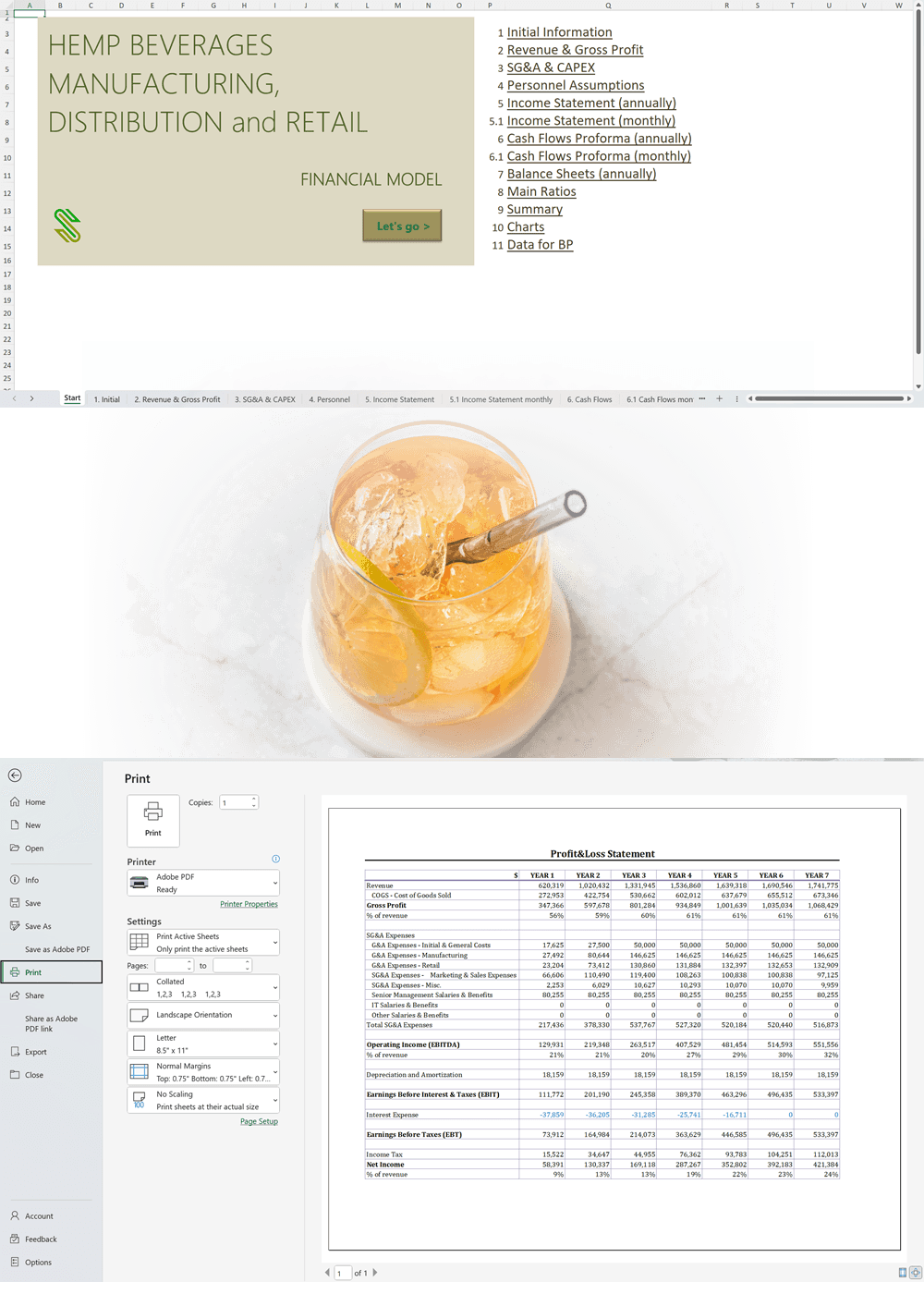
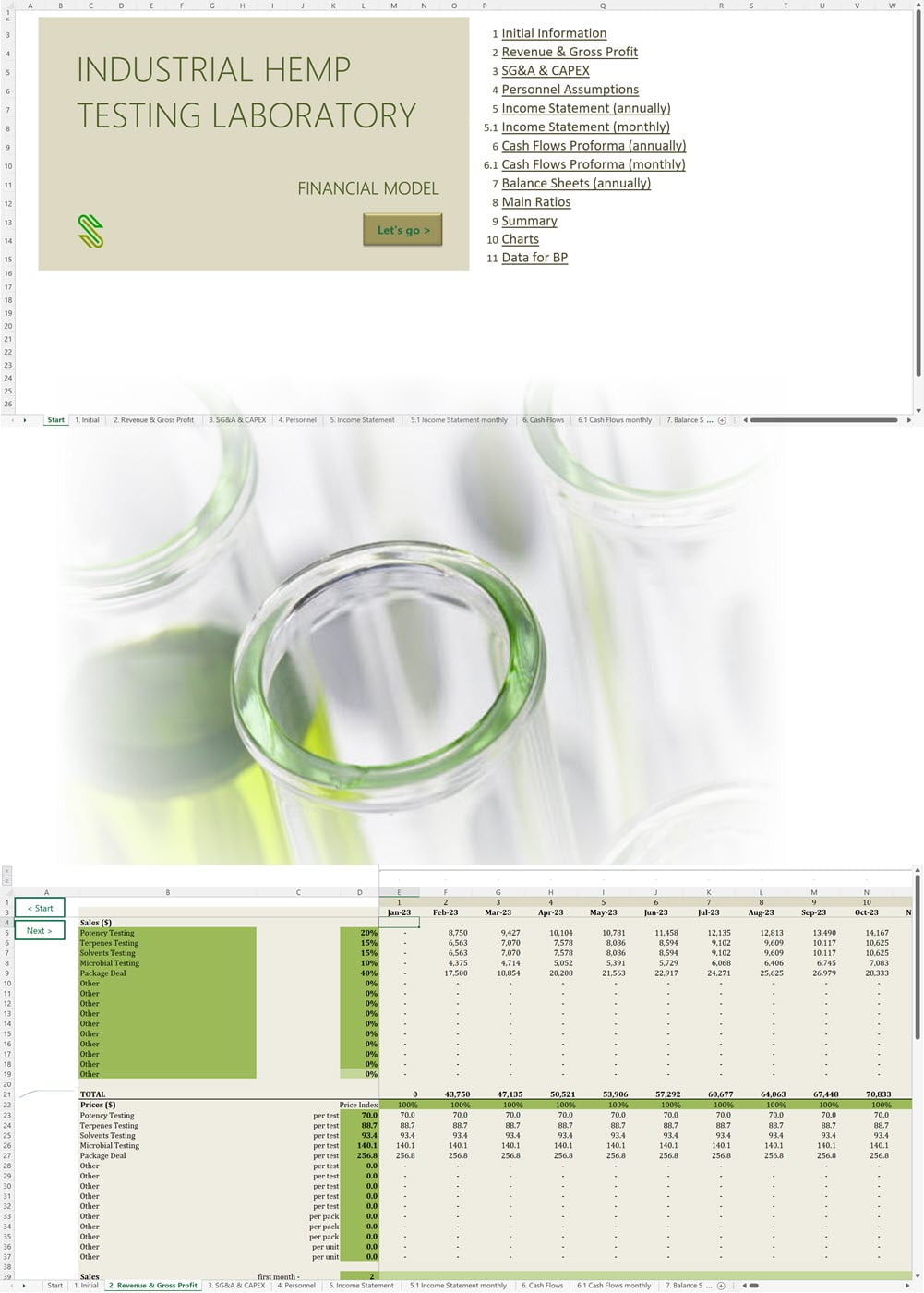
Business Plan Sample for a Hemp Cultivation and Processing Business in Uruguay
'70% ready to go' business plan templates
Our hemp/CBD financial models and business plan templates will help you estimate how much it costs to start and operate your own hemp/CBD business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Best Selling Templates
-

Hemp Cultivation Business Plan Template
Price range: $75.00 through $275.00 Select options This product has multiple variants. The options may be chosen on the product page -

Hemp CBD Financial Model All in One
$200.00 Select options This product has multiple variants. The options may be chosen on the product page -

CBD Retail Investor Pitch Deck Template
$75.00 Select options This product has multiple variants. The options may be chosen on the product page